Understanding that each patient is a real person beyond a number on a spreadsheet helps our team to focus on high quality and reliable sourcing of clinical trial supplies.
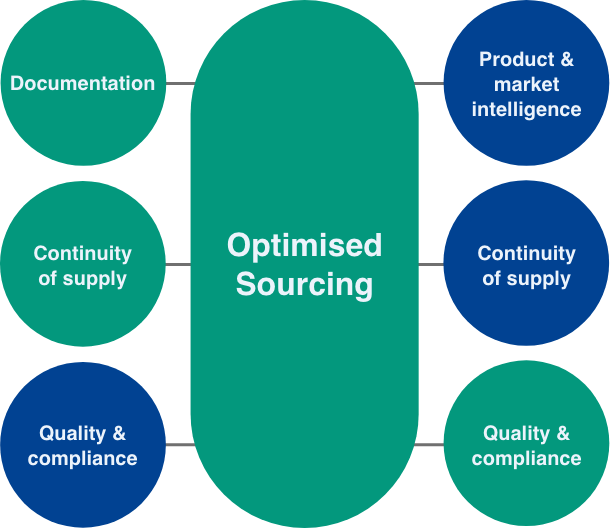
Reducing financial and supply chain risk to reliably deliver your clinical trial comparator and auxiliary medicines supply needs through data driven ethical sourcing practices.
We focus on reducing waste through our unique inventory reservation service and technology enabled sustainable supply chain processes, allowing you to focus on development of new treatments for patients.



Orifarm CTS sources from more than 27 global markets with over 20 years of experience
managing cross-country imports/exports.
With dedicated CTS teams and facilities in Denmark, UK, Czech Republic & USA we
can ensure the optimal supply chain routing to meet your requirements.


One size never fits all.
We view our customers as partners and take care to resource your projects with a
customised team to meet your specific needs